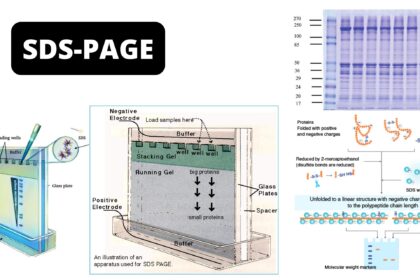
Introduction
Gel electrophoresis is a technique that separates molecules based on their size and charge by applying an electric current to a gel matrix. Basic steps of gel electrophoresis of DNA are:
- DNA is extracted.
- Isolation and amplification of DNA.
- DNA added to the gel wells.
- Electric current applied to the gel.
- DNA bands are separated by size.
- DNA bands are stained.
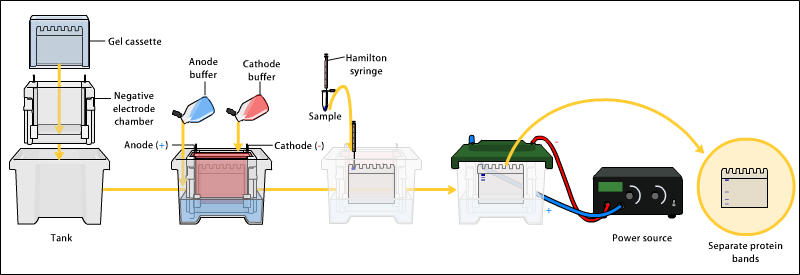
Applications of Gel Electrophoresis
Gel electrophoresis is widely used for separation and isolation of proteins and other macromolecules. It is an essential technique in molecular biology experiments, offering a range of analytical applications. It is also applied for distinguishing different species in DNA profiling for taxonomy studies, the study of structure and function of proteins, the analysis of antibiotic resistance, and the genetic similarity among populations or species1.
Applications in Molecular Biology
i) Determination of DNA sequences:
DNA sequencing relies on high resolution polyacrylamide gel electrophoresis. There are two methods currently in use, both based on generating radiolabeled fragments that terminate at specific bases. By using these fragments on the gel, it is possible to distinguish unique bands representing nucleotides and deduce the sequence.
ii) Southern and Northern blotting
Gel electrophoresis is a key technique used in DNA analysis. It allows us to determine whether a specific DNA sequence or mutation is present in a sample. By synthesizing a complementary radiolabeled oligonucleotide and exposing the sample to it, we can detect the desired sequence on a nitrocellulose paper using autoradiography. This technique is also applicable to RNA analysis in northern blotting.
iii) Restriction mapping of DNA:
During the study of genomic or cloned DNA it is essential to have some sort of map to differentiate one area from another. One of the techniques of mapping is known as restriction mapping. and the use of electrophoresis is. again. central to it. In its simplest form. The technique consists of :
- digesting the DNA with different restriction endonucleases.
- separating the resulting nucleotide fragments by agarose or sometimes polyacrylamide gel electrophoresis.
- visualizing these fragments. while-they are in the gel, by ethidium bromide staining and
- estimating their size in relation to the standards run on the gel simultaneously.
iv) Such important techniques as DNA footprinting (to find out the regions of DNA which interact with proteins) and restriction fragment length polymorphism (RFLP, used to detect mutations in various genes in carcinogenesis and other diseases) are dependent on gel electrophoresis in as much as that the final analysis is done on the gels after electrophoresis has been carried out.
(v) Among the most successful uses of analytical gels has been the detection of precursor molecules. which are processed (degraded) to give rise to the product mature species. The precursors differ very little in molecular weight from their products and the resolving power of the gel is the decisive factor in the experiment. Precursors of t-RNA. r-RNA and m-RNA have all been observed by their different mobilities on high-resolution gels.
(vi)The sensitivity of gel electrophoresis to variations in conformation of nucleic acids has not only been applied to separate nucleic acid molecules. But but also been used to study the kinetics of interconversions of conformation of many t-RNAs.
Applications in Protein Study
Determination of subunit stoichiometry
The subunit stoichiometry of an oligomeric protein might be determined by gel electrophoresis of the SDS-protein complex after covalent cross linkage has been introduced into the protein. The reagent dimethyl suberimidate NH = C (OCH3)-(CH2 )6-(CHP)C = NH is used to produce cross linkages between lysyl residues. This simple procedure is extensively used to determine the subunit stoichiometry of oligomeric proteins.
Determination of molecular weight of proteins
- SDS can be used to dissociate oligomeric proteins into their individual polypeptide chains. The degree of SDS-protein binding is very high, and thus the charge on the SDS-protein complex is almost entirely due to the exposed sulfate ions.
- The surface charge of this complex per unit area is constant regardless of the charge of the individual polypeptide chains.
- It has been demonstrated that such complexes tend to assume the shape of a long rod whose width is constant; the length of such complexes, therefore, becomes a function of the molecular weight of the polypeptide portion.
- These factors are at the base of molecular weight determination by SDS-gel electrophoresis.
- The method has gained extreme popularity because of its simplicity, reproducible results, and easy interpretation.
- Molecular weight of quite a few proteins has been determined by this method.
| Application | Purpose | Examples |
|---|---|---|
| DNA Electrophoresis | Separation of DNA fragments by size | DNA profiling, RFLP analysis, DNA sequencing |
| RNA Electrophoresis | Separation of RNA by size | RNA integrity analysis, Northern blotting |
| Protein Electrophoresis | Separation of proteins by size or charge | SDS-PAGE, Western blotting, IEF |
| Forensic Electrophoresis | DNA profiling for identification | Crime scene analysis, paternity testing |
| Clinical Electrophoresis | Disease diagnosis and monitoring | Hemoglobin electrophoresis, serum protein electrophoresis |
| Agarose Gel Electrophoresis | Separation of large DNA fragments | PCR analysis, plasmid examination |
| Polyacrylamide Gel Electrophoresis (PAGE) | High-resolution separation of biomolecules | Protein, RNA, and DNA analysis |
| Capillary Electrophoresis | High-resolution separation in capillaries | DNA/RNA/protein sequencing and analysis |
| 2D Gel Electrophoresis | Separation by charge and size | Comparative proteomics, PTM identification |
| Quality Control | Assess purity of biotech products | Nucleic acid, protein quality control |
Check out Introduction, types and principle of electrophoresis
References
- Alberts, B., Johnson, A., Lewis, J., Raff, M., Roberts, K., & Walter, P. (2002). Molecular Biology of the Cell (4th ed.). Garland Science.
- Sambrook, J., Fritsch, E. F., & Maniatis, T. (1989). Molecular Cloning: A Laboratory Manual (2nd ed.). Cold Spring Harbor Laboratory Press.
- Ausubel, F. M., Brent, R., Kingston, R. E., Moore, D. D., Seidman, J. G., Smith, J. A., & Struhl, K. (Eds.). (1995). Current Protocols in Molecular Biology. Wiley.
- Walker, J. M., & Rapley, R. (Eds.). (2009). Molecular Biomethods Handbook. Humana Press.
- Green, M. R., & Sambrook, J. (2017). Molecular Cloning: A Laboratory Manual (4th ed.). Cold Spring Harbor Laboratory Press.
- Maniatis, T., Fritsch, E. F., & Sambrook, J. (1982). Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press.
- Syaifudin, Mukh. (2021). Gel electrophoresis: The applications and its improvement with nuclear technology. AIP Conference Proceedings. 2331. 050008. 10.1063/5.0042067. ↩︎