Introduction
- PNK is a homotetramer with phosphatase activity at 3’ end and kinase activity at 5’ end with a tunnel like active site. The active site has side chains which interact with NTP donor’s beta-phosphate and 3’ phosphate of acceptor with an acid which activated 5’ –OH. Lys-15 and Ser-16 are important for the kinase activity of the enzyme.
- The basic residues of active site of PNK interact with the negatively charged phosphates of the DNA.
- Polynucleotide kinase (PNK) catalyzes the transfer of a phosphate group (PO4-2) from γ position of ATP to the 5′ end of either DNA or RNA and nucleoside monophosphate.
- PNK can convert 3′ PO4/5′ OH ends into 3′ PO4/5′ PO4 ends which blocks further ligation by ligase enzyme.
- T4 polynucleotide kinase is the most widely used PNK in molecular cloning experiments is isolated from T4 bacteriophage infected E.coli.
T4 Polynucleotide Kinase
T4 Polynucleotide Kinase (T4 PNK) catalyzes the transfer of the γ-phosphate from ATP to the 5´-terminus of polynucleotides or to mononucleotides bearing a 3´-phosphate group. T4 PNK is widely used to end-label short oligonucleotide probes, DNA and RNA molecules. Under certain conditions, the reaction with polynucleotides can be made reversible, permitting exchange of the γ-phosphate of ATP with the 5′ terminal phosphate of a polynucleotide, thus circumventing the need to dephosphorylate the substrate with alkaline phosphatase.
Principle: (PNK) T4 Polynucleotide Kinase catalyzes the transfer and exchange of the terminal gamma position phosphate of ATP to the 5′-hydroxyl terminus of double-and single-stranded DNA, RNA and nucleoside 3′-monophosphate molecules. T4 PNK also exhibits 3′-phosphatase and 2′, 3′-cyclicphosphodiesterase activities.
Features
- Incorporates labeled phosphates at the 5’ end of nucleic acids.
- Catalyzes transfer and exchange of phosphate to the 5′-OH of ssDNA and RNA.
- Catalyzes transfer and exchange of phosphate to the 5′-OH of dsDNA.
- Removes 3′ phosphoryl groups from DNA and RNA or a nucleotide/nucleoside.
PNK types of enzymatic activity
- Forward reaction: y-phosphate is transferred from ATP to the 5′ end of a polynucleotide (DNA or RNA). 5’ phosphate is not present either due to chemical synthesis or dephosphorylation. The 5’ OH nucleophile is activated by abstraction of the proton. Asp35 of PNK forms the co-ordinate bond with 5’ OH and attacks γ phosphorus forming an intermediate.
- Exchange reaction: target DNA or RNA having a 5′ phosphate is incubated with an excess of ADP – where PNK transfers the phosphate from the nucleic acid to an ADP, forming ATP. PNK then performs a forward reaction and transfer a phosphate from ATP to the target nucleic acid. Exchange reaction is used to label with radioactive phosphate group.
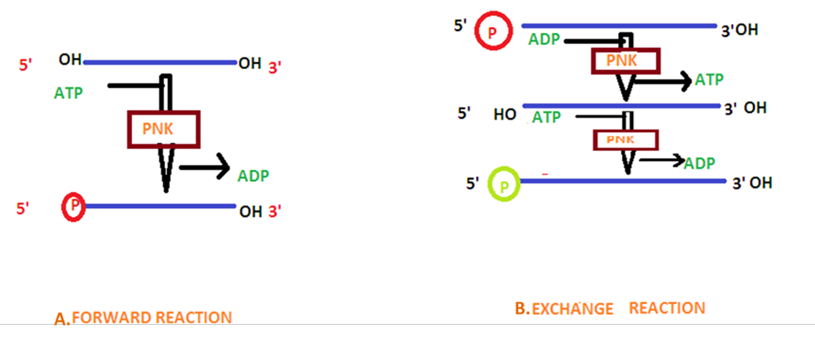
The efficiency of phosphorylation is less in exchange reaction compared to forward reaction. Along with the phosphorylating activity, PNK also has 3′ phosphatase activity.
Applications of PNK
- Labeling 5′-termini of nucleic acids to be used as:
- probes for hybridization,
- probes for transcript mapping,
- markers for gel-electrophoresis,
- primers for DNA sequencing,
- primers for PCR.
- 5′-phosphorylation of oligonucleotides, PCR products, other DNA or RNA prior to ligation.
- Phosphorylation of PCR primers.
- Detection of DNA modification by the [32P]-post labeling assay.
- Removal of 3′-phosphate groups (2)
Applications of T4 Polynucleotide Kinase in genetic engineering.
- 5′ phosphorylation of DNA/RNA for subsequent ligation
- End labeling DNA or RNA for probes and DNA sequencing
- Removal of 3′ phosphoryl groups
PNK is inhibited by ammonium ions, so ammonium acetate cannot be used to precipitate nucleic acids before phosphorylation. Sometimes phosphate ions or NaCl of greater than 50 mM concentration can also inhibit this enzyme.

